Research Finds Efficient Boron Neutron Capture Therapy For Brain Tumor With Novel Boron Carrier
A new boron agent drastically improves the effectiveness of boron neutron capture therapy for glioblastoma, demonstrate researchers at Tokyo Tech. The agent is selectively absorbed by brain tumor cells, exhibits enhanced blood retention, and can be administered at low doses. Experiments on cell cultures, mice, and rats show promising results, highlighting the potential of the novel agent for radiotherapy.
Glioblastoma (GBM) is a highly aggressive form of brain tumor that originates from supportive cells of the brain called glia. Due to its rapid growth, surgical removal of GBM is often difficult, leaving radiotherapy as the most viable option. Among the many existing radiotherapeutic modalities, boron neutron capture therapy (BNCT) has attracted a lot of attention for the treatment of GBM.
BNCT exploits the high affinity of boron-10 (10B) atoms for low-energy neutrons. When 10B absorbs a neutron, nuclear reactions release high-energy particles that damage the nearby biological tissue. Therefore, an essential requirement in BNCT is to achieve a significantly higher concentration of 10B in tumor cells than that in healthy normal cells. However, this has proven to be challenging, resulting in low survival rates and limited use of BNCT.
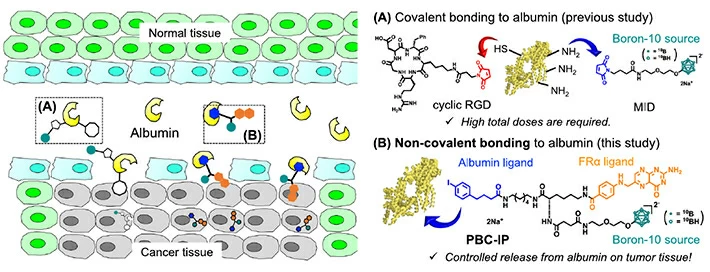
PBC-IP consists of three main functional groups (Figure 1): the first is a boron group containing twelve 10B atoms, the second is a ligand designed to bind to folate receptor α (FRα). This receptor, hardly present in normal cells, is greatly overexpressed in various cancers, including GBM. Thus, it acts as the entry point for PBC-IP into tumor cells. Finally, the third group is the 4-(p-iodophenyl)butyric acid moiety, which binds the entire molecule to albumin—an abundant carrier protein in blood that transports substances throughout the body. PBC-IP binds non-covalently to naturally present albumin, which allows it to directly interact with tumor cells, promoting its cellular uptake. Thus, the acid moiety can enhance the blood retention of the boron agent, thereby potentially reducing the required dose.
Figure 1 A new boron agent for the radiation-based treatment of glioblastoma. (A) Previously studied and (B) proposed boron agents used to deliver boron to tumor cells. The way in which the new boron agent PBC-IP binds to the carrier protein albumin facilitates its accumulation in tumor cells even at low doses, leading to better treatment outcomes without toxic effects on healthy cells.
The researchers conducted several experiments to confirm the viability of PBC-IP for BNCT, finding that it accumulated in GBM cell cultures 10–20 times more than L-4-boronophenylalanine (BPA), a clinically approved boron agent in Japan. In addition, PBC-IP showed no signs of toxicity to cells on its own, demonstrating its safety. “Likewise, PBC-IP administrated intravenously to the human GBM xenograft model showed higher boron accumulation in tumors than BPA, effectively suppressing tumor growth after thermal neutron irradiation,” highlights Prof. Nakamura.
Overall, the proposed boron agent may represent a breakthrough in radiotherapy for GBM, with the researchers currently conducting preclinical studies.