New Drug Reduces Stroke Damage in Mice
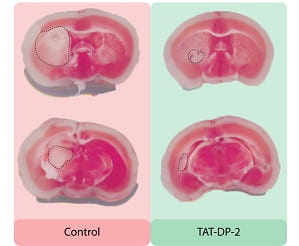 The study, published today in Science Advances, shows that injured neurons can remain viable if they are prevented from following biochemical pathways that lead to cell death.
The study, published today in Science Advances, shows that injured neurons can remain viable if they are prevented from following biochemical pathways that lead to cell death.
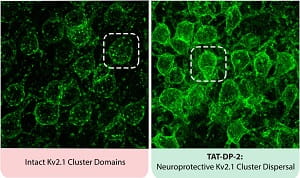 For this new study, the researchers identified two types of Kv2.1 channels in the neuronal cell membrane. One channel is routinely involved in cell excitability, while the other allows entry of additional, new Kv2.1 channels into the membrane, promoting the release of potassium and ultimately neuronal cell death.
For this new study, the researchers identified two types of Kv2.1 channels in the neuronal cell membrane. One channel is routinely involved in cell excitability, while the other allows entry of additional, new Kv2.1 channels into the membrane, promoting the release of potassium and ultimately neuronal cell death.

